Sign Up to our social questions and Answers Engine to ask questions, answer people’s questions, and connect with other people.
Login to our social questions & Answers Engine to ask questions answer people’s questions & connect with other people.
Lost your password? Please enter your email address. You will receive a link and will create a new password via email.
Please briefly explain why you feel this question should be reported.
Please briefly explain why you feel this answer should be reported.
Please briefly explain why you feel this user should be reported.
Suppose that a = 2.3 cm. Determine the magnitude of the electric field at point…

See lessConstants An idealized ammeter is connected to a battery as shown in the figure

See lessIt is the responsibility of the _____ workstation to contact the patient about an Action…
1. The production station must contact the patient to inform them of an Action Note before picking up. 2. Otic preparations can be placed in the ear. 3. The NDC number for the medication stock bottle is 11 digits. 4. Directions for taking the medication is NOT typically provided on a medication stocRead more
1. The production station must contact the patient to inform them of an Action Note before picking up.
2. Otic preparations can be placed in the ear.
3. The NDC number for the medication stock bottle is 11 digits.
4. Directions for taking the medication is NOT typically provided on a medication stock bottle.
5. Prescription is being partially filled.
6. RxNet does not have Employee Personnel Records.
7. Only a pharmacist can manage Schedule II (CII), according to CVS policy.
8. The Auxillary sticker is located on the top of the prescription label. It will indicate whether the customer requested non safety caps. Also, it will indicate if they will need to sign the Acknowledgment for CVS’ Notice Privacy Practices.
9. The Poison Prevention Packaging Act is overseen by the Customer Product Safety Commission.
10. A syringe is used to administer sterile preparations.
11. We request the patient’s acknowledgement that she has received our Notice of Privacy Policies (NoPP) when a patient files their first prescription at any CVS.
12. State board of pharmacy agency licenses Pharmacy employees
13. The customer is responsible for reviewing completed Action Notes at the pick-up desk and communicating any remaining information to them.
14. Ointments can be directly applied to the skin.
15. Tablets are taken by mouth
See lessFind the critical value Za/2 that corresponds to the given confidence level

See lessThe graph below shows the voltage and current graphs for an inductor.
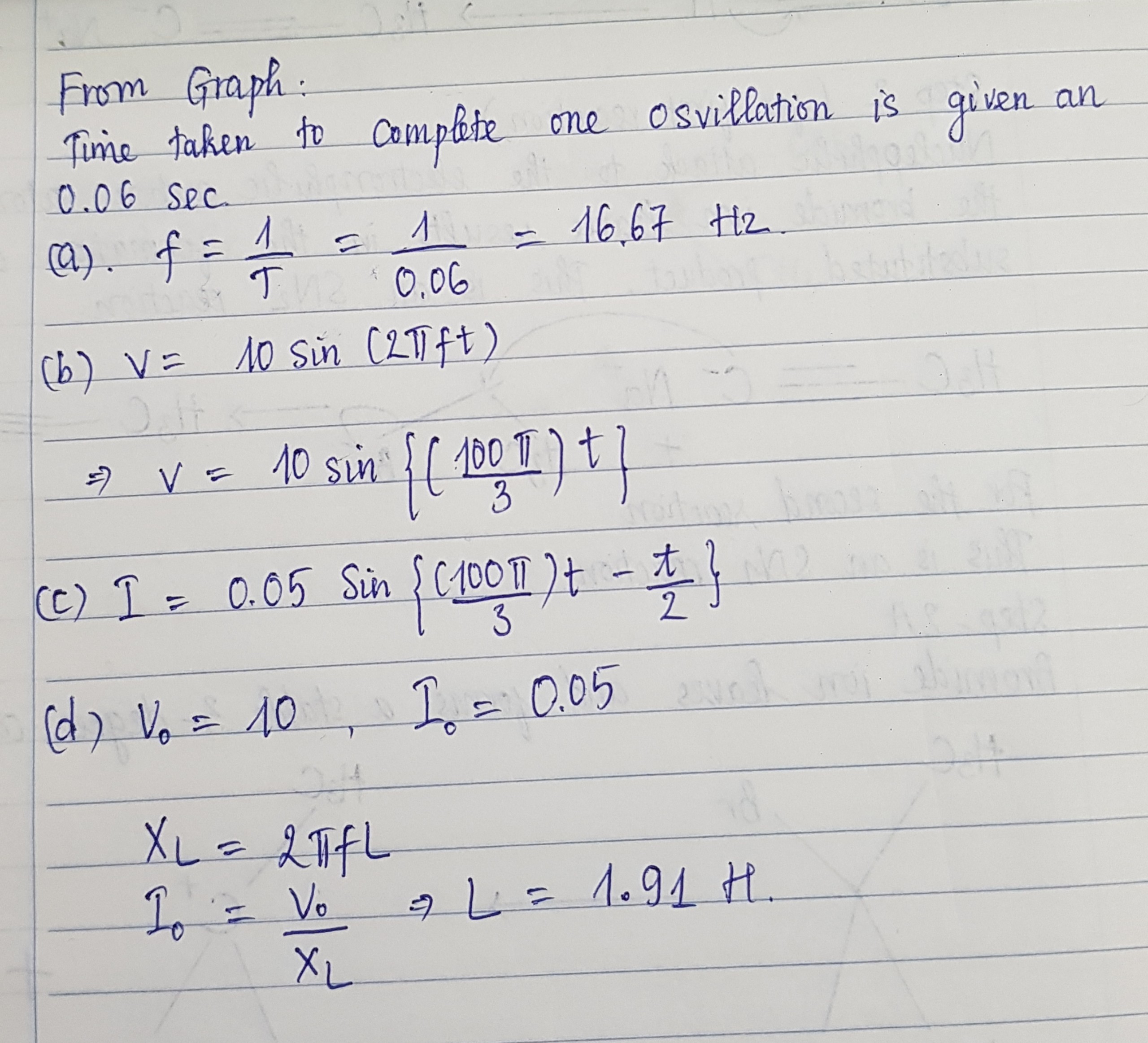
See lessThe earth has a radius of 6380 km and turns around once on its axis in 24h.
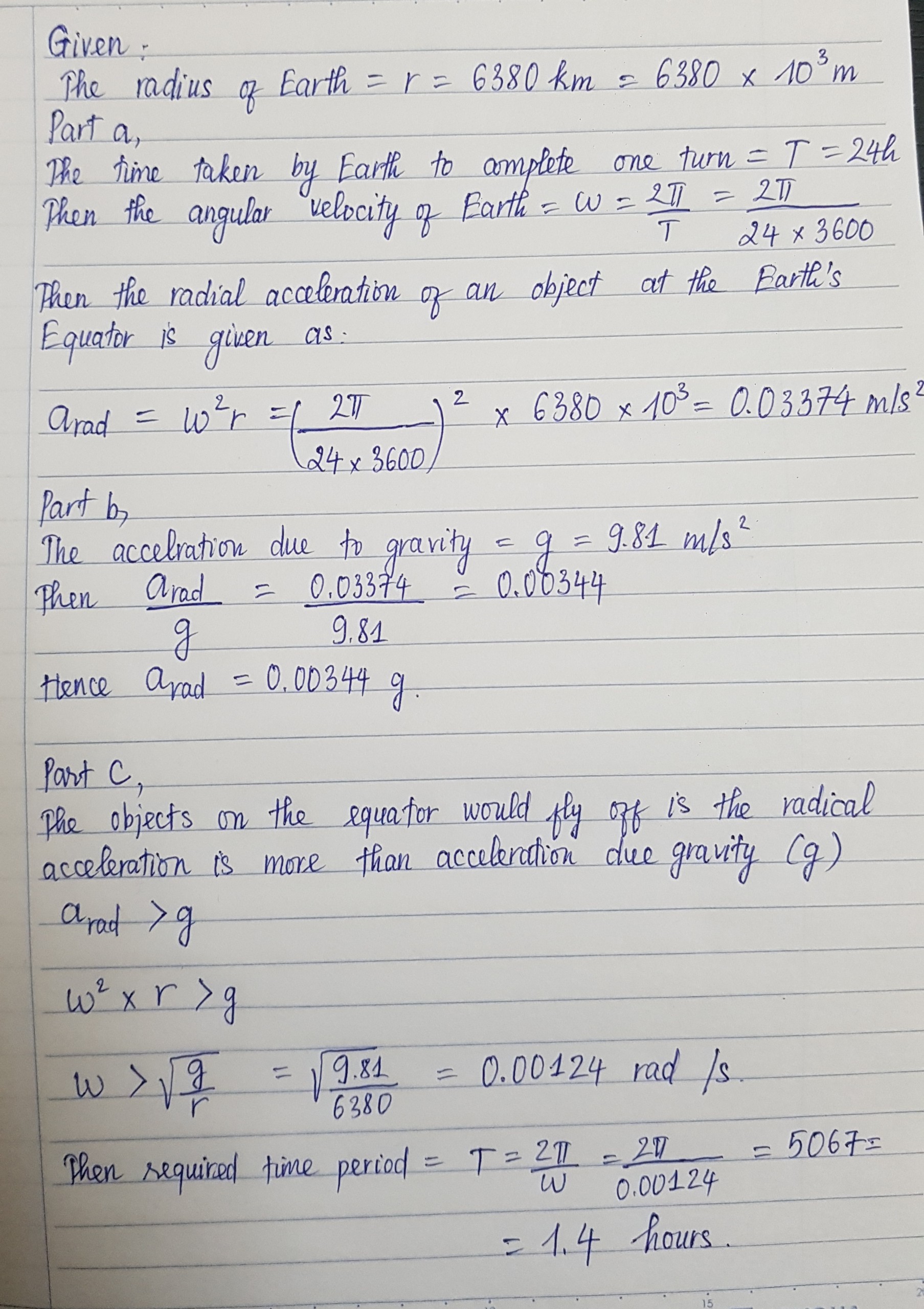
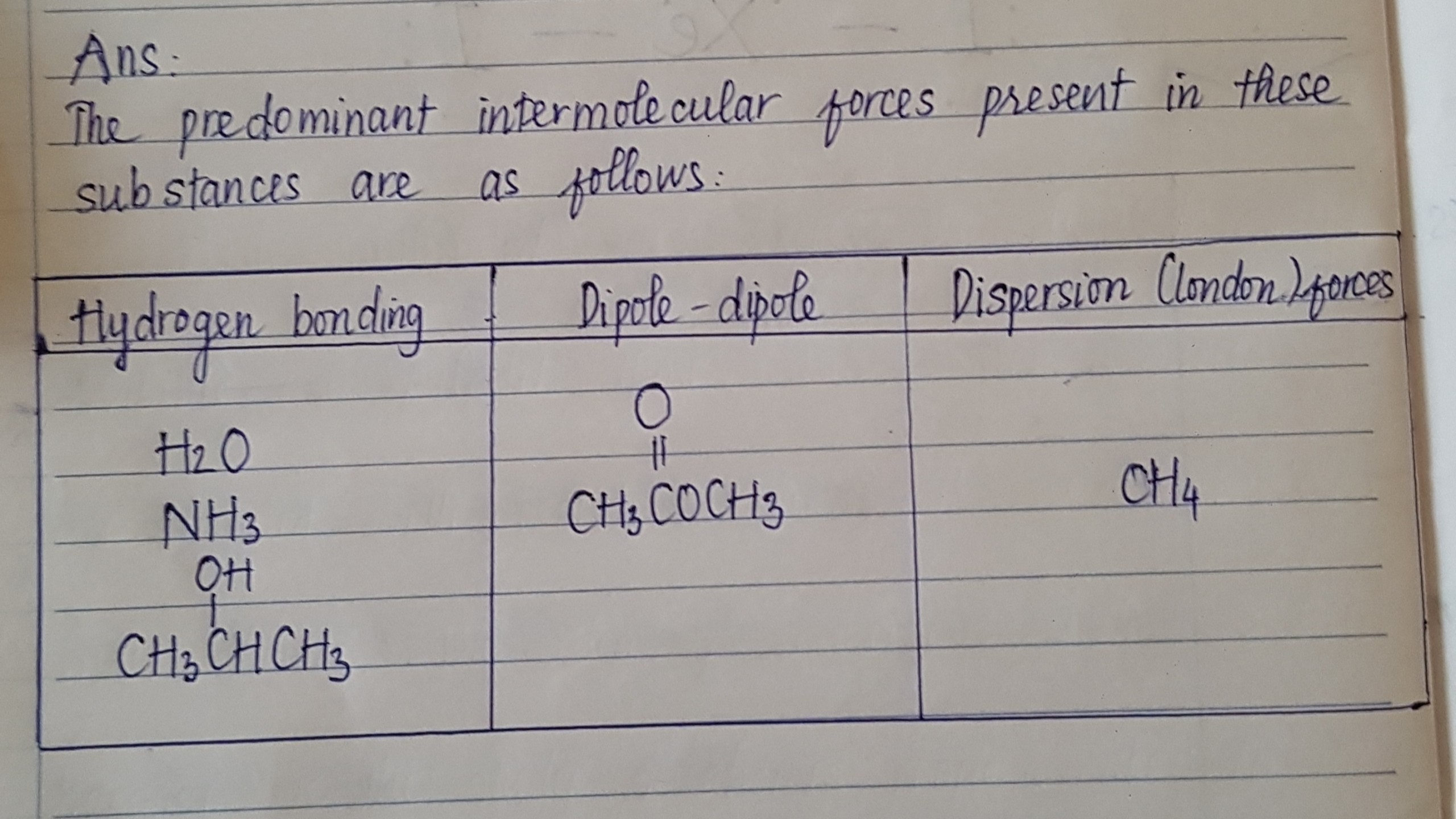
See lessIdentify the predominant intermolecular force in each of these substances.
Concepts and Reason Attractive forces develop between the nucleus and electrons of two atoms, or molecules, when they approach one another. A change in electron distribution occurs within a molecule. Intermolecular forces refers to interactions between neighboring molecules and atoms. Fundamentals LRead more
Attractive forces develop between the nucleus and electrons of two atoms, or molecules, when they approach one another. A change in electron distribution occurs within a molecule. Intermolecular forces refers to interactions between neighboring molecules and atoms.
London-dispersion forces, which are weak and exhibited all the molecules, are not strong.
Hydrogen bonding can be strong and is demonstrated by molecules that have O-H, N-H or F-H bond.
The dipole-dipole interactions between molecules with net dipoles are mild and are exhibited by them.
These compounds are:
CH3COOCH3 has dispersion forces and dipole-dipole interactions
H2O, NH3, and C3H7OH has hydrogen bonding because of bonds. They also have dipole/dipole interactions as well as dispersion forces.
CH4 only dispersion forces exist.
This is the order in which intermolecular forces are ranked:
CH3COOCH3 is therefore dominated by dipole-dipole interactions.
Hydrogen bonding is the most common interaction in .
.
The most common interactions in CH4 are London or dispersion interactions.
See lessThe wire shown in Fig. below is infinitely long and carries a current I.

See lessDraw the best Lewis structure for XeI2.
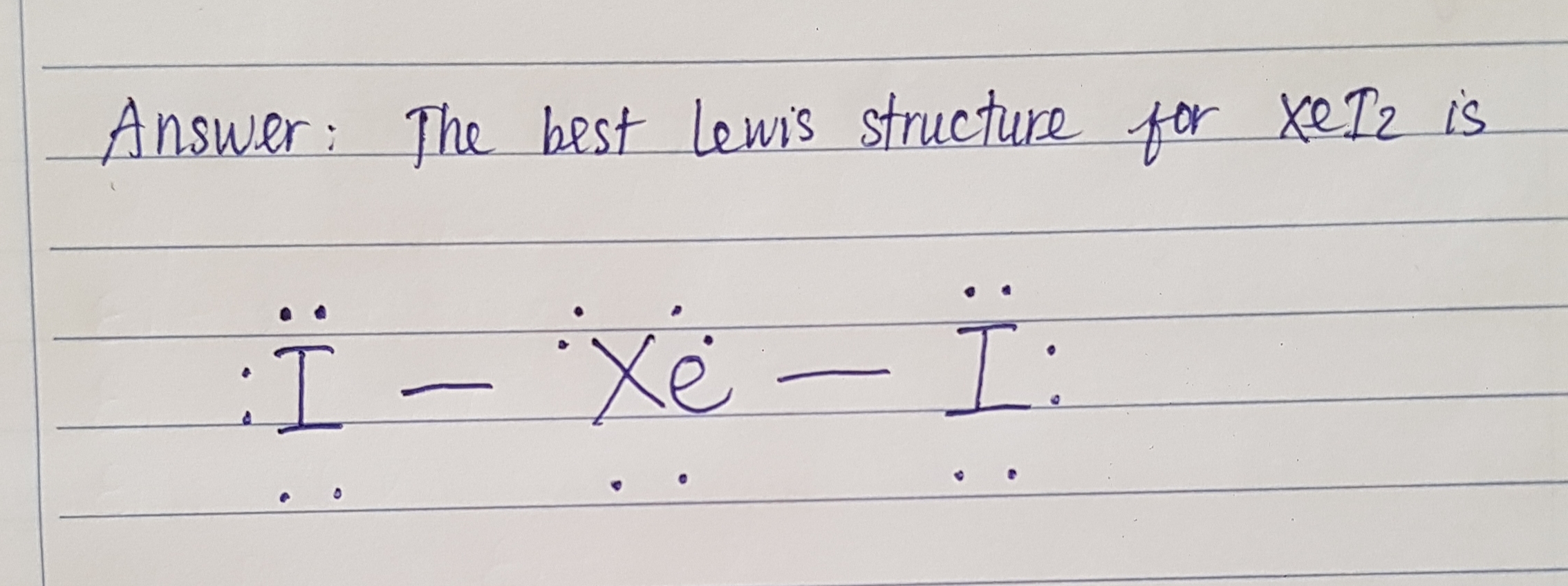
See lessWhat is the electric flux through the cylinder due to this infinite line of charge?

See less